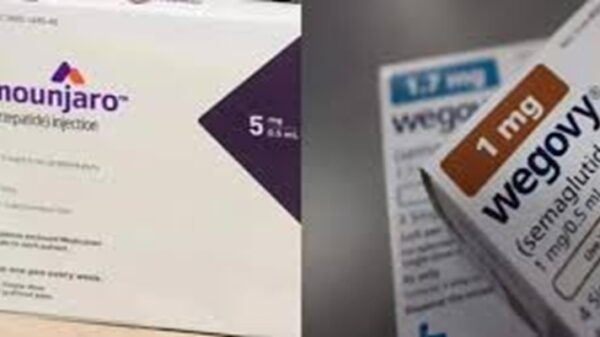
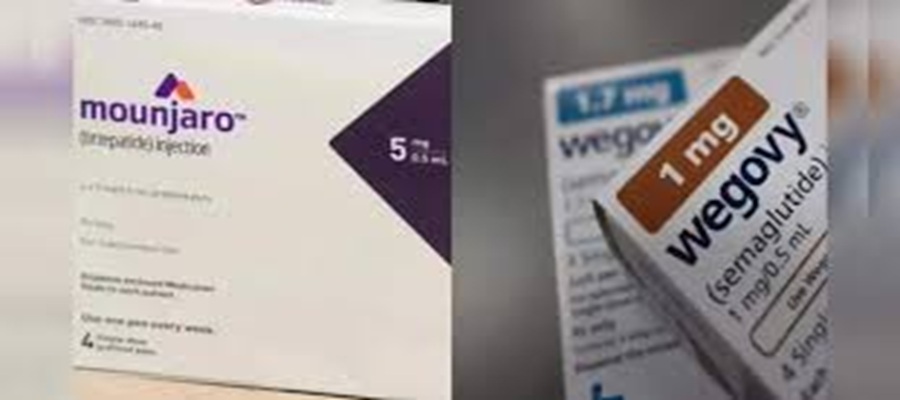
The US FDA proposed on April 30, 2026, to exclude key ingredients like semaglutide (Ozempic, Wegovy), tirzepatide (Mounjaro, Zepbound), and liraglutide from the 503B bulk substances list used by outsourcing facilities for compounding.
Commissioner Marty Makary emphasized that with FDA-approved versions available, there’s no clinical need for bulk compounding outside shortages, aiming to protect patients from risks like dosage errors and poor quality control.
Compounded drugs, unlike FDA-approved generics, skip full agency verification despite using approved ingredients; prior tolerance stemmed from demand surges but ended as shortages eased.
If finalized after public comments due June 29, compounding resumes only during verified shortages, boosting Novo Nordisk shares over 6% and Eli Lilly over 8%. This upholds drug approval integrity while allowing science-based exceptions.
![]()